
National Sun Yat-sen University / Associate Prof. Wen-Fan Chen
Currently, large segmental bone defects are still primarily treated with autografts and allografts, yet both approaches present significant limitations. Autologous bone offers the most complete regenerative capacity and excellent biocompatibility, but its availability is limited, harvesting requires damage to healthy bone, and patients face risks of donor-site pain and infection. As a result, autografts are impractical for large defects.
Allografts are more readily available, yet may trigger immune rejection or disease transmission, and their use remains constrained by the limited supply of bone banks. In addition, most commercial synthetic bone substitutes on the market, typically composed of HA, TCP, or similar materials, provide only osteoconductivity. Their intrinsic mechanical strength is insufficient, so they are commonly used in powder, granule, or paste form, making them incapable of withstanding load-bearing conditions and unsuitable to replace autografts or metallic implants in long bones, the spine, or weight-bearing sites.
To date, there is still no synthetic bone substitute that simultaneously combines high mechanical strength, bioactivity, and degradability. Consequently, when clinicians encounter extensive bone defects, reconstruction of load-bearing segments, or the need for patient-specific implants, there remains a lack of safe and effective non-metallic alternatives.
Technology Introduction
The “high-strength 3D-printed biomimetic ceramic bone substitute” developed in this technology uses a highly biocompatible biomimetic ceramic as the core material. It exhibits a compressive strength of up to 1.25 GPa, surpassing conventional HA/TCP and even medical-grade titanium alloys. Moreover, it possesses controllable degradability, allowing gradual replacement by new bone while maintaining mechanical support during healing.
The material demonstrates excellent biocompatibility and osteoinductive activity, promoting cell adhesion, bone ingrowth, and new bone formation. Animal studies have shown that its bone repair rate is three times faster than commercial HA-based products. By integrating DLP photopolymerization 3D printing with a specialized sintering process, it enables fabrication of bioactive implants with precise pore architectures and fully customized geometries.
Depending on the defect morphology, it can be manufactured into bone plates, bone struts, interbody fusion cages, or craniofacial reconstruction implants. Combining high mechanical strength, bone regenerative capability, and patient-specific customization, this technology has strong potential to replace metallic and polylactic acid–based implants, reduce secondary surgeries, and provide a safer and more effective biomedical solution for bone defect treatment.
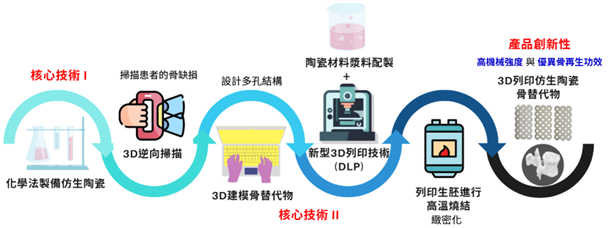
Figure 1. Production Workflow of High-Strength 3D-Printed Biomimetic Ceramic Bone Substitutes
Application Examples
Reconstruction of long bone defect:
Customized high-strength scaffolds support load-bearing segments and gradually degrade, allowing new bone to replace the implant over time.
Spinal fusion implants:
3D-printed interbody fusion cages promote bone ingrowth and vertebral fusion, eliminating the need for secondary removal surgery.
Craniofacial bone reconstruction:
Implants are printed according to defect geometry, achieving both anatomical restoration and bone regeneration.
Core decompression surgery implants:
3D-printed porous scaffolds fill the void, maintain mechanical integrity, and promote bone ingrowth and healing.
Post-tumor resection reconstruction:
For large segmental defects, patient-specific scaffolds provide structural support while facilitating new bone regeneration.
Related Links
None
Patent Name and Number
曾郁升,陳玟帆:玻璃陶瓷及其製造方法,中華民國專利,發明第I883553號,2025/5/11-2043/9/20
Industry-Academia / Tech Transfer Partner
None
Honors and Awards
None
Technical Contact
Industrial Liaison Office, National Sun Yat-sen University
Tel: +886 7-5250165
Email: gloria@mail.nsysu.edu.tw