﹤Biotech & Biomed Innovation﹥Technologies for Solid-Phase Peptide Ligation
Kaohsiung Medical University / Prof. Chai-Lin Kao, Hui-Ting Chen etc.
Pain Points Solved
- Conventional solid-phase peptide synthesis often suffers from incomplete reactions, low product purity, and unstable yields when producing long-chain or difficult peptides.
- Existing ligation technologies are limited in reaction efficiency, resulting in lengthy processes, high material consumption, and elevated manufacturing costs.
- Insufficient quality consistency during scale-up production hinders the development and commercialization of pharmaceutical peptide products.
Technology Introduction
Our team applies the advantages of solid-phase synthesis to develop an innovative patented ligation technology that enhances synthetic efficiency and product purity, maximizing yield. This technology significantly reduces material costs, laborious effort, and processing time while enabling the synthesis of peptides exceeding 40-mers. Our approach is instrumental in both the production of generic peptide drugs and the development of novel peptide derivatives. Remarkably, this method could apply to all dimensions of peptides, including linear, cyclic, and branched peptides, amphiphilic peptides, and peptide-drug conjugates. Those are difficult to produce through conventional methods. By optimizing synthetic efficiency and scalability, our technology offers a cost-effective solution for peptide manufacturing, meeting the demands of pharmaceutical and biotechnological applications.
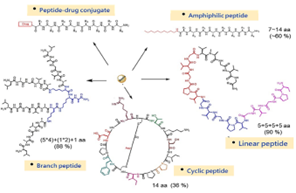
Figure. Types of synthetic peptides that utilized our patented technologies.
Application Examples
- Enables high-efficiency synthesis of long-chain peptides exceeding 40 amino acids, supporting rapid development and production of generic peptide drugs.
- Applicable to complex peptide architectures that are difficult to produce using conventional methods, including cyclic, branched, amphiphilic peptides, and peptide–drug conjugates.
- Helps biotechnology and pharmaceutical companies reduce production costs while improving scalability and economic feasibility of peptide manufacturing.
Related Links
None
Patent Name and Number
TW I772159、TW I480070 、TW I535467 、TW 113139108、TW 114113738
US 18/558,364、US 8,889,828、US 9,314,533、US 9,415,114
EP 21954443.4
PCT/CN 2024/124690、PCT/CN 2025/088393
Industry-Academia / Tech Transfer Partner
None
Honors and Awards
22nd National Innovation Award-Academia & Research Innovation Award
Technical Contact
Mr. Hung, Assistant Manager
Kaohsiung Medical University
Tel: +886 7-3121101 ext. 2360
Email: R121084@kmu.edu.tw